15. Chemical Kinetics
Reaction Mechanism
Problem 112a
Textbook Question
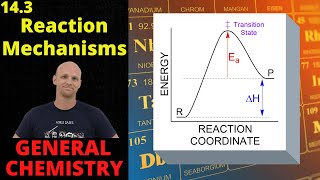
Textbook QuestionIn a hydrocarbon solution, the gold compound 1CH323AuPH3 decomposes into ethane 1C2H62 and a different gold compound, 1CH32AuPH3. The following mechanism has been proposed for the decomposition of 1CH323AuPH3: Step 1: 1CH323 AuPH3 Δ k1 k -1 1CH323Au + PH3 1fast2 Step 2: 1CH323 Au ¡k2 C2H6 + 1CH32Au 1slow2 Step 3: 1CH32Au + PH3 ¡k3 1CH32AuPH3 1fast2 (c) What is the molecularity of each of the elementary steps?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
355
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos