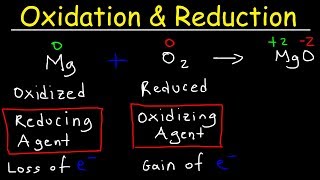
20. Electrochemistry
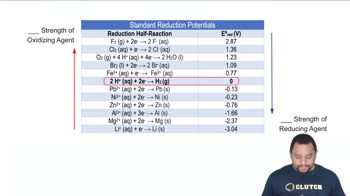
Standard Reduction Potentials
Multiple Choice
Multiple ChoiceIn the following reaction identify the oxidizing agent and the reducing agent:
Cr2O72- + 6 Fe2+ + 14 H+ → 2 Cr3+ + 6 Fe3+ + 7 H2O
A
Oxidizing Agent:Fe, Reducing Agent:H
B
Oxidizing Agent:Fe, Reducing Agent:Cr
C
Oxidizing Agent:Cr, Reducing Agent:Fe
D
Oxidizing Agent:H, Reducing Agent:Fe
924
views
7
rank
5
comments
Related Videos
Related Practice
Showing 1 of 10 videos