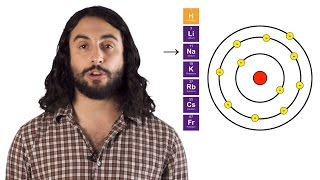
10. Periodic Properties of the Elements
Periodic Trend: Ionization Energy
Problem 103
Textbook Question
Textbook QuestionConsider the electronic structure of the element bismuth. (d) Would you expect element 115 to have an ionization ene-rgy greater than, equal to, or less than that of bismuth? Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
443
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos