18. Aqueous Equilibrium
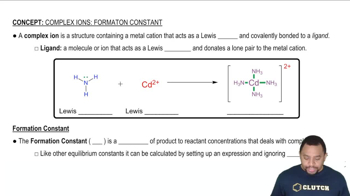
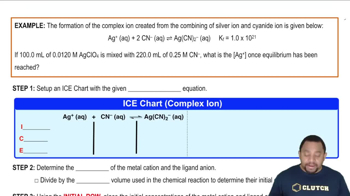
Complex Ions: Formation Constant
Multiple Choice
Multiple ChoiceIf your equilibrium constant K is equal to the product of Ksp and Kf, find the solubility of AgCl in 2.0 M NH3. Ksp of AgCl = 1.77 × 10−10; Kf of Ag(NH3)2+ = 1.7 × 107.
A
1.04 × 10−17 M
B
9.78 × 10−3 M
C
9.89 × 10−2 M
D
3.01 × 10−3 M
189
views
1
rank
1
comments
Related Videos
Related Practice
Showing 1 of 2 videos