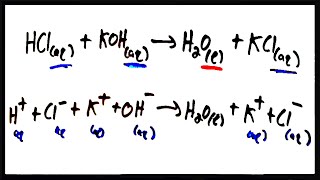6. Chemical Quantities & Aqueous Reactions
Molecular Equations
Problem 88
Textbook Question
Textbook QuestionAssume that you have an aqueous solution of an unknown salt. Treatment of the solution with dilute NaOH, Na2SO4, and KCl produces no precipitate. Which of the following cations might the solution contain? (a) Ag+ (b) Cs+ (c) Ba2+ (d) NH4 +
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
639
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos