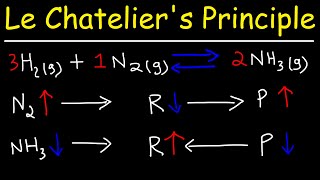
16. Chemical Equilibrium
Le Chatelier's Principle
Problem 143
Textbook Question
Textbook QuestionGiven the Arrhenius equation, k = Ae-Ea>RT, and the rela- tion between the equilibrium constant and the forward and reverse rate constants, Kc = kf>kr, explain why Kc for an exothermic reaction decreases with increasing temperature.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
406
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos