8. Thermochemistry
Heat Capacity
Problem 46
Textbook Question
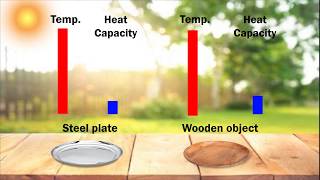
Textbook QuestionA kilogram of aluminum metal and a kilogram of water are each warmed to 75 °C and placed in two identical insulated containers. One hour later, the two containers are opened and the temperature of each substance is measured. The aluminum has cooled to 35 °C, while the water has cooled only to 66 °C. Explain this difference.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
34sPlay a video:
1212
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos