15. Chemical Kinetics
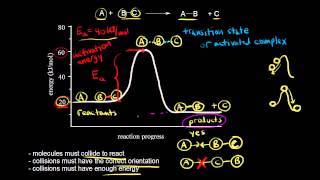
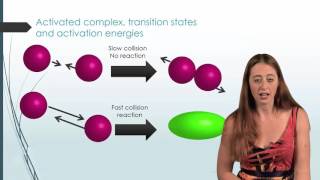
Collision Theory
Problem 59
Textbook Question
Textbook QuestionBased on their activation energies and energy changes and assuming that all collision factors are the same, rank the following reactions from slowest to fastest. (a) Ea = 45 kJ>mol; E = -25 kJ>mol (b) Ea = 35 kJ>mol; E = -10 kJ>mol (c) Ea = 55 kJ>mol; E = 10 kJ>mol
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1396
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos