20. Electrochemistry
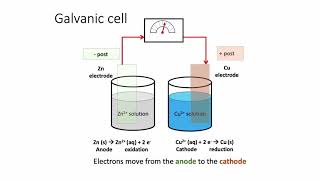
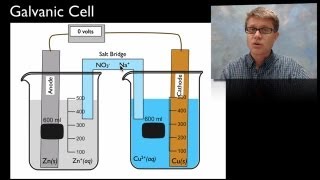
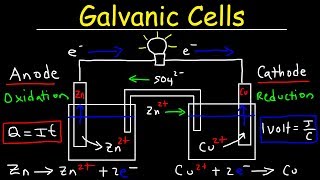
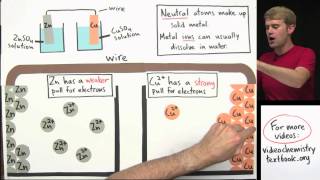
Galvanic Cell
Problem 42
Textbook Question
Textbook QuestionA voltaic cell consists of a strip of cadmium metal in a solution of Cd1NO322 in one beaker, and in the other beaker a platinum electrode is immersed in a NaCl solution, with Cl2 gas bubbled around the electrode. A salt bridge connects the two beakers. (c) Write the equation for the overall cell reaction.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
1148
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos