12. Molecular Shapes & Valence Bond Theory
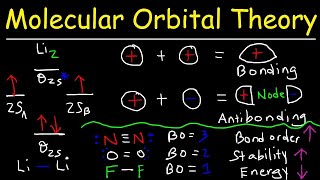
Molecular Orbital Theory
Problem 104
Textbook Question
Textbook QuestionThe highest occupied molecular orbital of a molecule is abbreviated as the HOMO. The lowest unoccupied molecular orbital in a molecule is called the LUMO. Experimentally, one can measure the difference in energy between the HOMO and LUMO by taking the electronic absorption (UV-visible) spectrum of the molecule. Peaks in the electronic absorption spectrum can be labeled as p2p9p2p*, s2s9s2s*, and so on, corresponding to electrons being promoted from one orbital to another. The HOMO-LUMO transition corresponds to molecules going from their ground state to their first excited state. (c) The electronic absorption spectrum of the N2 molecule has the lowest energy peak at 170 nm. To what orbital transition does this correspond?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
2863
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos