8. Thermochemistry
Constant-Volume Calorimetry
Problem 59b
Textbook Question
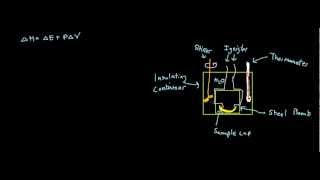
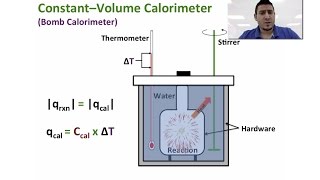
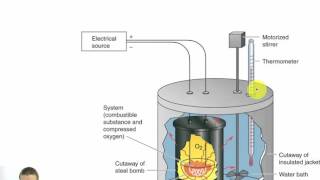
Textbook QuestionUnder constant-volume conditions, the heat of combustion of benzoic acid (C6H5O6) is 15.57 kJ/g. A 3.500-g sample of sucrose is burned in a bomb calorimeter. The temperature of the calorimeter increases from 20.94 to 24.72 °C. (b) If the size of the sucrose sample had been exactly twice as large, what would the temperature change of the calorimeter have been?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1026
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos