20. Electrochemistry
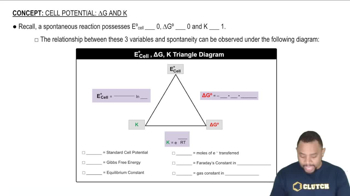
Cell Potential: ∆G and K
Problem 160
Textbook Question
Textbook QuestionThe nickel–iron battery has an iron anode, an NiO(OH) cathode, and a KOH electrolyte. This battery uses the follow-ing half-reactions and has an E° value of 1.37 V at 25 °C. (b) Calculate ∆G° (in kilojoules) and the equilibrium con-stant K for the cell reaction at 25 °C.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
995
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos