13. Liquids, Solids & Intermolecular Forces
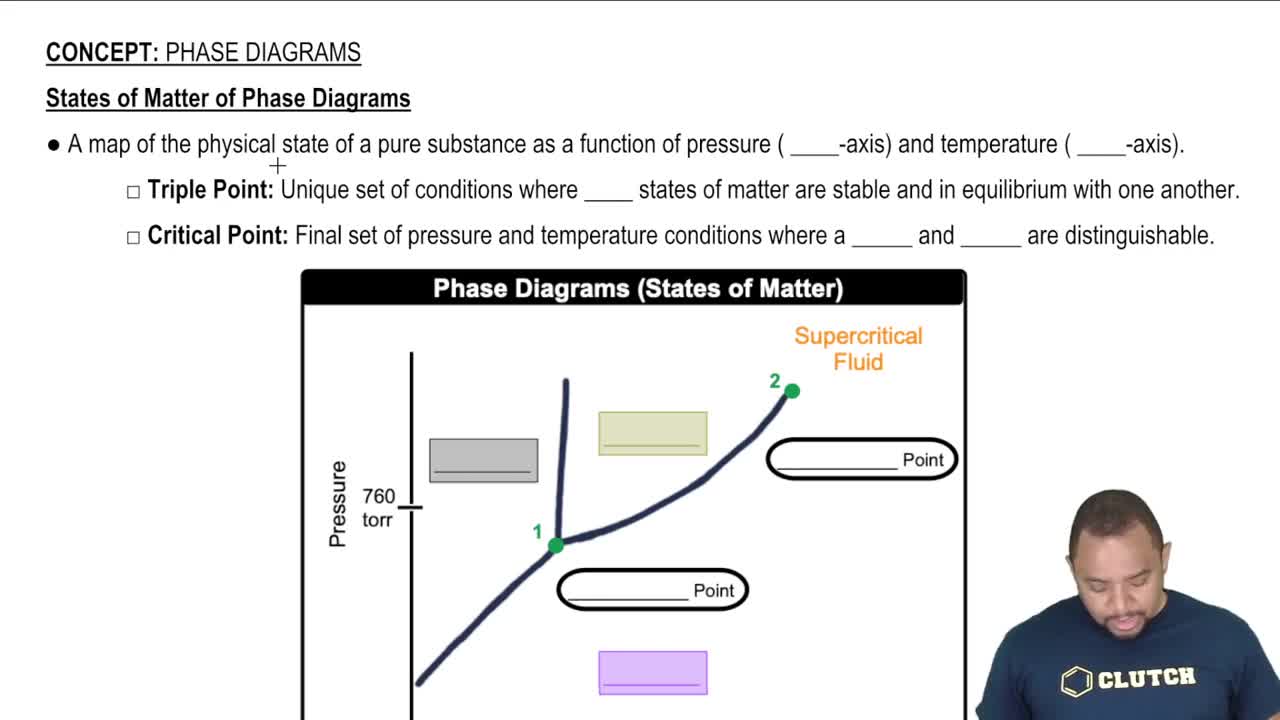
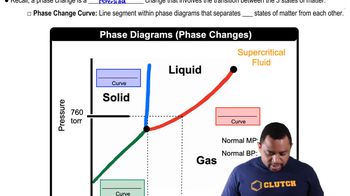
Phase Diagrams
Problem 87
Textbook Question
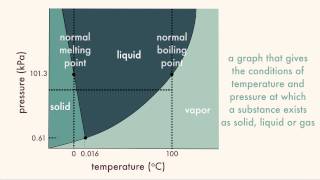
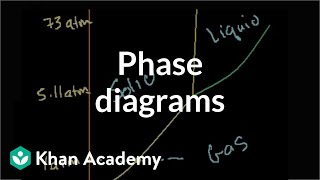
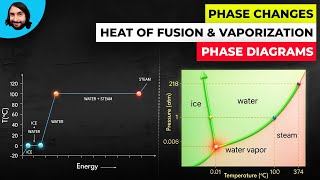
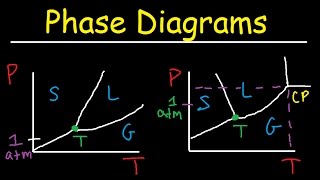
Textbook QuestionNaphthalene (C10H8) is the main ingredient in traditional mothballs. Its normal melting point is 81 °C, its normal boiling point is 218 °C, and its triple point is 80 °C at 1000 Pa. Using the data, construct a phase diagram for naphthalene, labeling all the regions of your diagram.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
2373
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos