3. Chemical Reactions
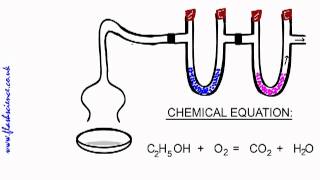
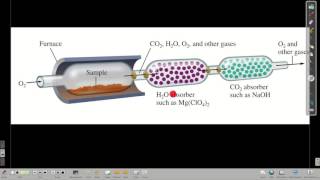
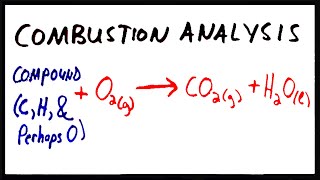
Combustion Analysis
Multiple Choice
Multiple ChoiceA compound composed of carbon, hydrogen and nitrogen undergoes a combustion reaction to produce 264.21 g CO2, 63.06 g H2O and 46.4 g NO2. Determine its empirical formula.
A
C2H11N3
B
C6H7N
C
C6H12N4
D
CHN
317
views
5
rank
Related Videos
Related Practice
Showing 1 of 9 videos