12. Molecular Shapes & Valence Bond Theory
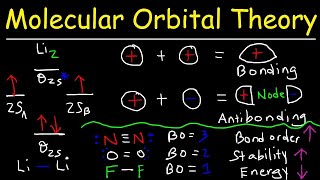
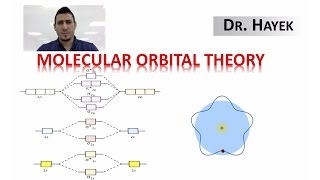
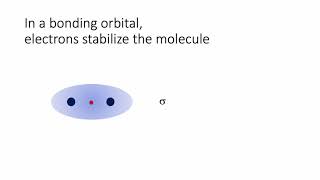
Molecular Orbital Theory
Problem 6
Textbook Question
Textbook QuestionThe orbital diagram that follows presents the final step in the formation of hybrid orbitals by a silicon atom. (a) Which of the following best describes what took place before the step pictured in the diagram: (i) Two 3p electrons became unpaired, (ii) An electron was promoted from the 2p orbital to the 3s orbital, or (iii) An electron was promoted from the 3s orbital to the 3p orbital?

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
605
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos