20. Electrochemistry
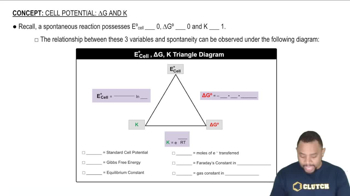
Cell Potential: ∆G and K
Problem 52c
Textbook Question
Textbook QuestionFor each of the following reactions, write a balanced equation, calculate the standard emf, calculate ∆G° at 298 K, and calculate the equilibrium constant K at 298 K. (b) In acidic solution, copper(I) ion is oxidized to copper(II) ion by nitrate ion.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
10mPlay a video:
372
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos