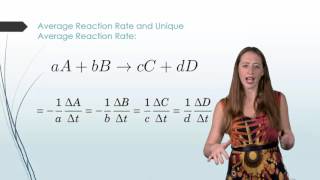15. Chemical Kinetics
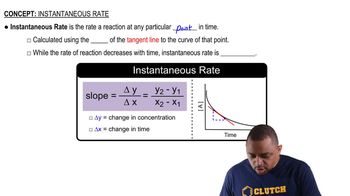
Instantaneous Rate
Problem 33a
Textbook Question
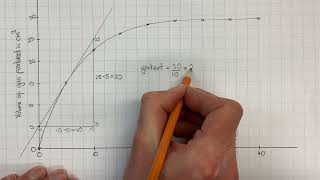
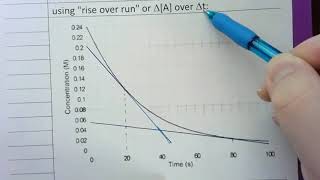
Textbook QuestionConsider the reaction: H2( g) + Br2( g) ¡ 2 HBr( g) The graph shows the concentration of Br2 as a function of time. a. Use the graph to calculate each quantity: (iii) the instantaneous rate of formation of HBr at 50 s
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
907
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos