24. Transition Metals and Coordination Compounds
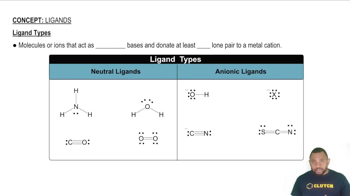
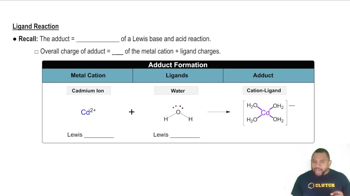
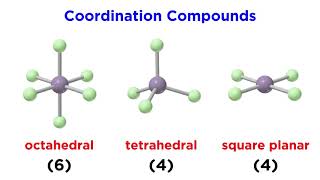
Ligands
Problem 3
Open Question
Open QuestionWhat is the Lewis base in the reaction of oxalate with the
mangenese ion to form [Mn(C2O4)3]2-? What is the oxidation state of Mn and the coordination number of the complex?
(a) Lewis base is C2O42-; Mn oxidation number is +3; coordination number is 3.
(b) Lewis base is C2O42-; Mn oxidination number is +2; coordination number is 6.
(c) Lewis base is Mn2+; Mn oxidation number is +2; coordination number is 3.
(d) Lewis base is Mn4+; Mn oxidation number is +4; coordination number is 6.
332
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos