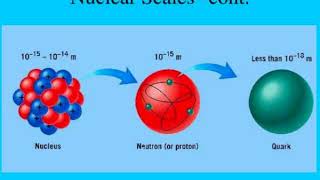
21. Nuclear Chemistry
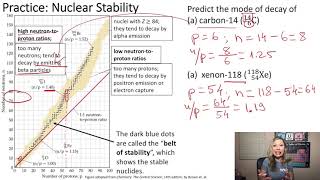
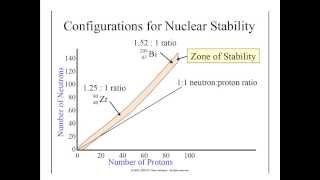
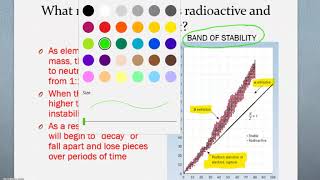
Neutron to Proton Ratio
Problem 1
Textbook Question
Textbook QuestionIndicate whether each of the following nuclides lies within the belt of stability in Figure 21.2: (a) neon-24. For any that do not, describe a nuclear decay process that would alter the neutron-to-proton ratio in the direction of increased stability. [Section 21.2]

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
1367
views
1
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos