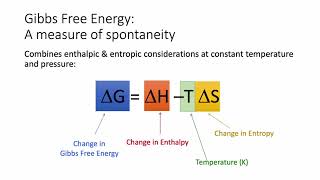
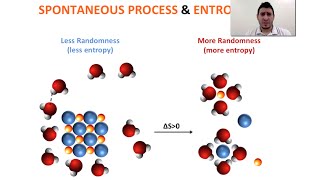
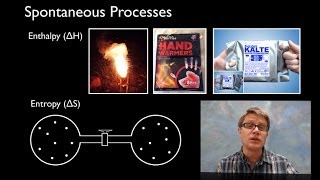
19. Chemical Thermodynamics
Spontaneous vs Nonspontaneous Reactions
Multiple Choice
Multiple ChoiceA positive ΔSsystem indicates what about the overall spontaneity of a process?
A
The process is always spontaneous.
B
The process is always nonspontaneous.
C
ΔSsurroundings is negligible.
D
ΔSsurroundings must also be positive.
E
Nothing; only ΔSuniverse can tell about the overall spontaneity of a process.
222
views
Related Videos
Related Practice
Showing 1 of 9 videos