17. Acid and Base Equilibrium
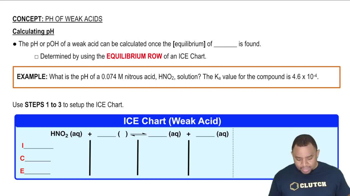
pH of Weak Acids
Problem 147
Textbook Question
Textbook QuestionWhen NO2 is bubbled into water, it is completely converted to HNO3 and HNO2: 2 NO21g2 + H2O1l2S HNO31aq2 + HNO21aq2 Calculate the pH and the concentrations of all species present (H3O+ , OH-, HNO2, NO2 -, and NO3 -) in a solution prepared by dissolving 0.0500 mol of NO2 in 1.00 L of water. Ka for HNO2 is 4.5 * 10-4.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
10mPlay a video:
966
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 5 videos