17. Acid and Base Equilibrium
pH of Weak Bases
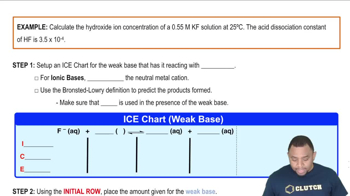
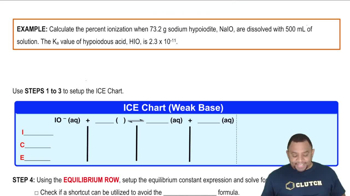
Problem 73
Textbook Question
Textbook QuestionEphedrine, a central nervous system stimulant, is used in nasal sprays as a decongestant. This compound is a weak organic base: C10H15ON1aq2 + H2O1l2 Δ C10H15ONH+1aq2 + OH-1aq2 A 0.035 M solution of ephedrine has a pH of 11.33. (b) Calculate Kb for ephedrine.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
457
views
Was this helpful?
Related Videos
Related Practice