3. Chemical Reactions
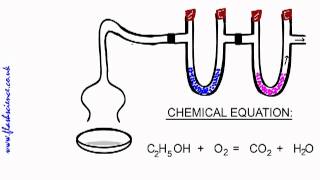
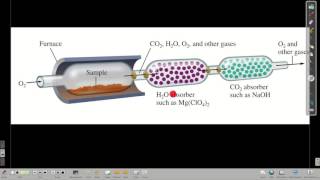
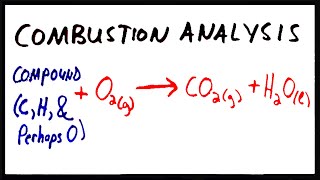
Combustion Analysis
Open Question
Open QuestionThe combustion of 4.16 grams of a compound which contains only C,H,O and F yields 7.7 g CO2 and 2.52 g H2O. Another sample of the compound with a mass of 3.63 g is found to contain 0.58 g F. What is the empirical formula of the compound?
 Verified Solution
Verified SolutionVideo duration:
7mPlay a video:
1325
views
13
rank
3
comments
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos