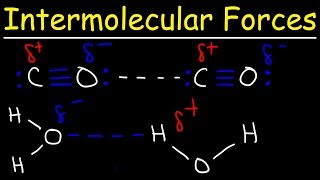
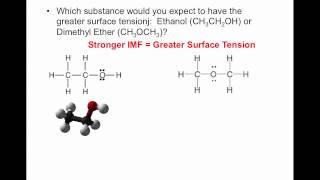
13. Liquids, Solids & Intermolecular Forces
Intermolecular Forces and Physical Properties
Problem 32
Textbook Question
Textbook QuestionThe generic structural formula for a 1-alkyl-3-methylimid- azolium cation is where R is a -CH2(CH2)nCH3 alkyl group. The melting points of the salts that form between 1-alkyl-3-methylimidazolium cation and the PF6- anion are as follows: R = CH2CH3 (m.p. = 60 °C), R = CH2CH2CH3 (m.p. = 40 °C), r = CH2CH2CH2CH3 (m.p. = 10 °C), and R = CH2CH2CH2CH2CH2CH3 (m.p. = -61 °C). Why does the melting point decrease as the length of alkyl group increases?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
380
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos