11. Bonding & Molecular Structure
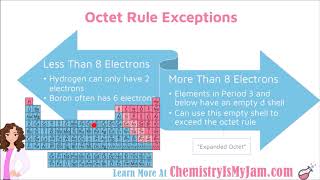
Lewis Dot Structures: Exceptions
Multiple Choice
Multiple ChoiceWhich of the following is false?
A
Molecules with an odd number of electrons are called radicals.
B
It is possible for stable compounds to have less than an octet of electrons.
C
Elements in period three and below (n < 3) may expand their octets into the d-subshell.
D
Elements in period three have energetically accessible d orbitals.
E
All of the above are true.
236
views
Related Videos
Related Practice
Showing 1 of 10 videos