13. Liquids, Solids & Intermolecular Forces
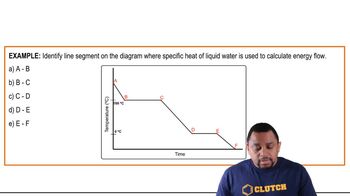
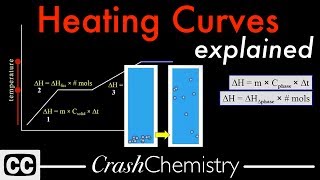
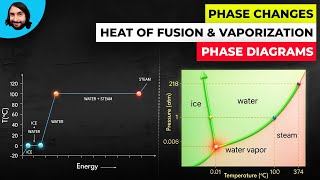
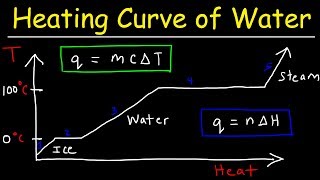
Heating and Cooling Curves
Problem 59
Textbook Question
Textbook QuestionSuppose that 0.95 g of water condenses on a 75.0-g block of iron that is initially at 22 °C. If the heat released during condensation goes only to warming the iron block, what is the final temperature (in °C) of the iron block? (Assume a constant enthalpy of vaporization for water of 44.0 kJ/mol.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
4548
views
5
rank
2
comments
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos