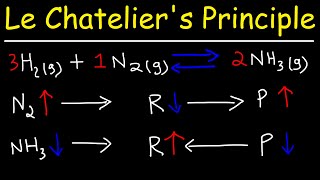
16. Chemical Equilibrium
Le Chatelier's Principle
Multiple Choice
Multiple ChoiceAt 445 ºC calculate the equilibrium concentration of HI when 0.500 M HI, 0.0200 M H2, and 0.0200 M I2 are placed in a sealed flask and allowed to reach equilibrium. Kc for this reaction is 50 at 445 ºC.
H2 (g) + I2 (g) ⇆ 2 HI (g)
A
0.020 M
B
0.060 M
C
0.460 M
D
0.421 M
E
0.579 M
292
views
Related Videos
Related Practice
Showing 1 of 13 videos