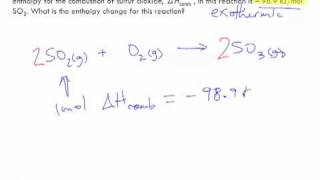8. Thermochemistry
Thermochemical Equations
Problem 115
Textbook Question
Textbook QuestionA gaseous fuel mixture contains 25.3% methane (CH4), 38.2% ethane (C2H6), and the rest propane (C3H8) by volume. When the fuel mixture contained in a 1.55 L tank, stored at 755 mmHg and 298 K, undergoes complete combustion, how much heat is emitted? (Assume that the water produced by the combustion is in the gaseous state.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
18mPlay a video:
2908
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos