18. Aqueous Equilibrium
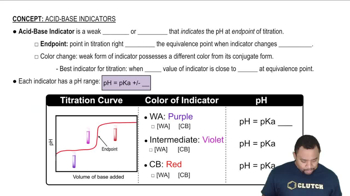
Acid-Base Indicators
Problem 148
Textbook Question
Textbook QuestionA 40.0 mL sample of a mixture of HCl and H3PO4 was titrated with 0.100 M NaOH. The first equivalence point was reached after 88.0 mL of base, and the second equiva-lence point was reached after 126.4 mL of base. (f) What indicators would you select to signal the equiva-lence points?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
561
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos