18. Aqueous Equilibrium
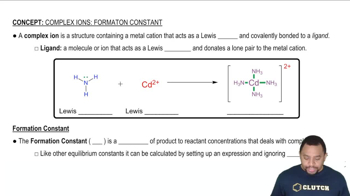
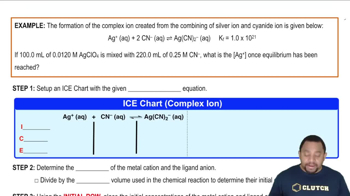
Complex Ions: Formation Constant
Problem 126
Textbook Question
Textbook QuestionCitric acid (H3Cit) can be used as a household cleaning agent to dissolve rust stains. The rust, represented as Fe(OH)3, dissolves because the citrate ion forms a soluble complex with Fe3+ (a) Using the equilibrium constants in Appendix C and Kf = 6.3 x 10^11 for Fe(Cit), calculate the equilibrium constant K for the reaction. (b) Calculate the molar solubility of Fe(OH)3 in 0.500 M solution of H3Cit.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
10mPlay a video:
465
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos