10. Periodic Properties of the Elements
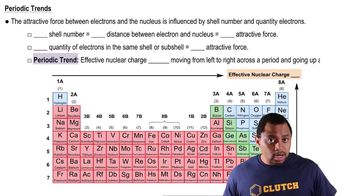
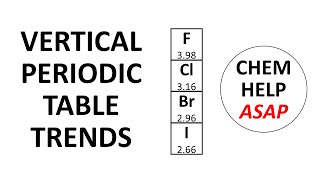
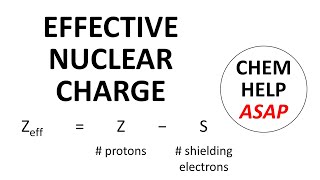
Periodic Trend: Effective Nuclear Charge
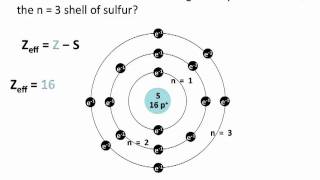
Problem 129
Textbook Question
Textbook QuestionOne method for calculating Zeff is to use the equation

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
999
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos