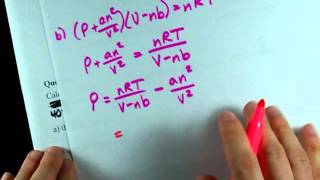7. Gases
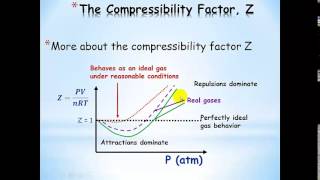
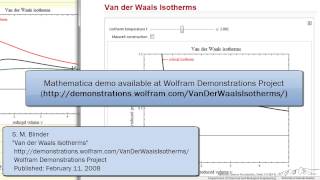
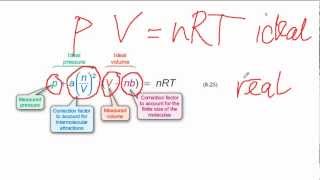
Van der Waals Equation
Problem 93
Textbook Question
Textbook QuestionUse the van der Waals equation and the ideal gas equation to calculate the volume of 1.000 mol of neon at a pressure of 500.0 atm and a temperature of 355.0 K. Explain why the two values are different. (Hint: One way to solve the van der Waals equation for V is to use successive approximations. Use the ideal gas law to get a preliminary estimate for V.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
3689
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos