2. Atoms & Elements
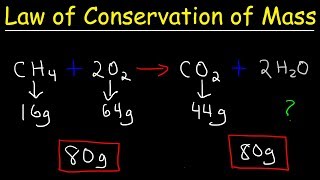
Law of Conservation of Mass
Problem 78
Textbook Question
Textbook QuestionA sample of mercury with a mass of 114.0 g was combined with 12.8 g of oxygen gas, and the resulting reaction gave 123.1 g of mercury(II) oxide. How much oxygen was left over after the reaction was complete?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
734
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos