18. Aqueous Equilibrium
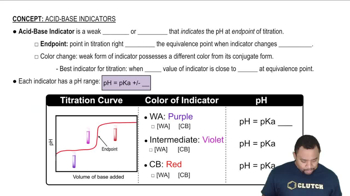
Acid-Base Indicators
Multiple Choice
Multiple ChoiceChemistry student is using an indicator with a pKa of 4.7 for the titration of a strong acid with strong base. Calculate the pH range at which the indicator will change colors.
a) 2.7–3.5 b) 10–11 c) 4.7–8.7 d) 3.7–5.7 e) 2.7–5.8
A
2.7–3.5
B
10–11
C
4.7–8.7
D
3.7–5.7
E
2.7–5.8
187
views
Related Videos
Related Practice
Showing 1 of 2 videos