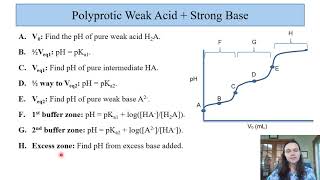
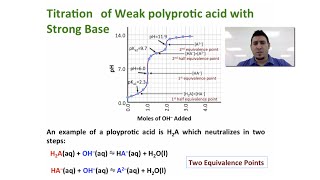
18. Aqueous Equilibrium
Titrations: Diprotic & Polyprotic Buffers
Multiple Choice
Multiple ChoiceSuppose you have 50.1 mL of a H3PO4 solution that you titrate with 15.4 mL of 0.10 M KOH solution to reach the endpoint. What is the concentration of H3PO4 of the original H3PO4 solution?
A
0.00200 M
B
0.00154 M
C
0.00649 M
D
0.0307 M
175
views
Related Videos
Related Practice
Showing 1 of 10 videos