18. Aqueous Equilibrium
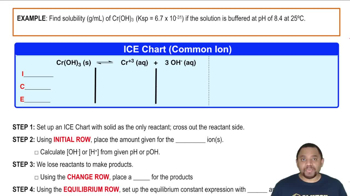
Ksp: Common Ion Effect
Problem 59
Textbook Question
Textbook QuestionConsider a beaker containing a saturated solution of CaF2 in equilibrium with undissolved CaF21s2. Solid CaCl2 is then added to the solution. (a) Will the amount of solid CaF2 at the bottom of the beaker increase, decrease, or remain the same?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1157
views
Was this helpful?