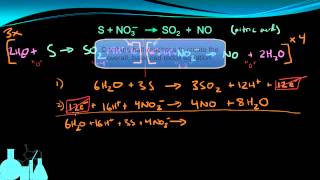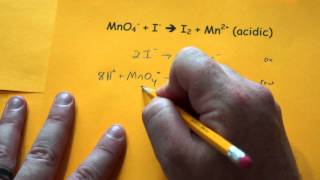6. Chemical Quantities & Aqueous Reactions
Balancing Redox Reactions: Acidic Solutions
Problem 155b
Textbook Question
Textbook QuestionSodium nitrite, NaNO2, is frequently added to processed meats as a preservative. The amount of nitrite ion in a sample can be determined by acidifying to form nitrous acid (HNO2), letting the nitrous acid react with an excess of iodide ion, and then titrating the I3 - ion that results with thiosulfate solution in the presence of a starch indicator. The unbalanced equations are (1) (2) (a) Balance the two redox equations.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
11mPlay a video:
570
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos