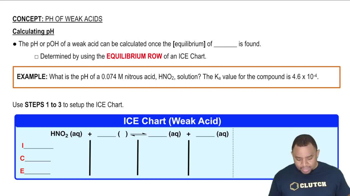
17. Acid and Base Equilibrium
pH of Weak Acids
Problem 31
Textbook Question
Textbook QuestionCalculate the percent ionization of a 0.15 M benzoic acid solution in pure water and in a solution containing 0.10 M sodium benzoate. Why does the percent ionization differ significantly in the two solutions?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
1967
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 5 videos