16. Chemical Equilibrium
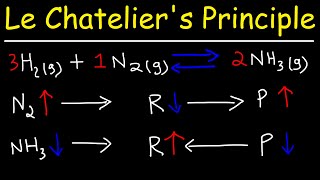
Le Chatelier's Principle
Problem 67b
Textbook Question
Textbook QuestionEach reaction is allowed to come to equilibrium, and then the volume is changed as indicated. Predict the effect (shift right, shift left, or no effect) of the indicated volume change. b. 2 H2S( g) ⇌ 2 H2( g) + S2( g) (volume is decreased)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1204
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos