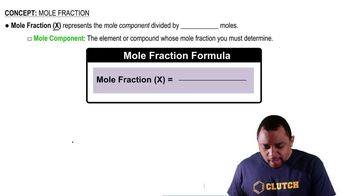
Fluorocarbons (compounds that contain both carbon and fluorine) were, until recently, used as refrigerants. The compounds listed in the following table are all gases at 25 °C, and their solubilities in water at 25 °C and 1 atm fluorocarbon pressure are given as mass percentages. (c) Infants born with severe respiratory problems are sometimes given liquid ventilation: They breathe a liquid that can dissolve more oxygen than air can hold. One of these liquids is a fluorinated compound, CF3(CF2)7Br. The solubility of oxygen in this liquid is 66 mL O2 per 100 mL liquid. In contrast, air is 21% oxygen by volume. Calculate the moles of O 2 present in an infant's lungs (volume: 15 mL) if the infant takes a full breath of air compared to taking a full 'breath' of a saturated solution of O 2 in the fluorinated liquid. Assume a pressure of 1 atm in the lungs. Fluorocarbon Solubility (mass %) CF4 0.0015 CClF3 0.009 CCl2F2 0.028 CHClF2 0.30
 Verified Solution
Verified Solution