10. Periodic Properties of the Elements
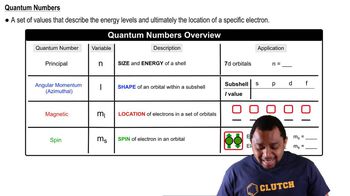
The Electron Configuration: Quantum Numbers
Multiple Choice
Multiple ChoiceGive the set of four quantum numbers that represent the indicated electron in each of the following elements.
Cl– (18th electron)
A
n = 3; l = 0; ml = +1; ms = -1/2
B
n = 3; l = 0; ml = -1; ms = -1/2
C
n = 3; l = 2; ml = +1; ms = -1/2
D
n = 3; l = 1; ml = +1; ms = +1/2
E
n = 3; l = 1; ml = +1; ms = -1/2
1504
views
2
rank
1
comments
Related Videos
Related Practice
Showing 1 of 5 videos