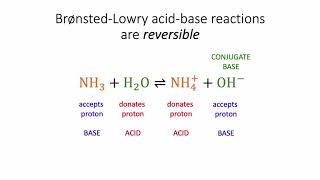17. Acid and Base Equilibrium
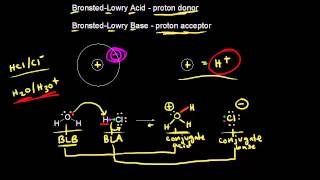
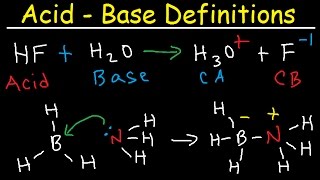
Bronsted-Lowry Acids and Bases
Multiple Choice
Multiple ChoiceIn the following reaction, label Bronsted-Lowry acid and base, along with conjugate acid and base.


H2C6H6O6 (aq) + H2O (l) ⇌ HC6H6O6− (aq) + H3O (aq)


A
H2C6H6O6 conjugate acid
H2O conjugate base
HC6H6O6− base
H3O+ acid
H2O conjugate base
HC6H6O6− base
H3O+ acid
B
H2C6H6O6 acid
H2O base
HC6H6O6− conjugate base
H3O+ conjugate acid
H2O base
HC6H6O6− conjugate base
H3O+ conjugate acid
C
H2C6H6O6 conjugate base
H2O conjugate acid
HC6H6O6− acid
H3O+ base
H2O conjugate acid
HC6H6O6− acid
H3O+ base
D
H2C6H6O6 base
H2O acid
HC6H6O6− conjugate acid
H3O+ conjugate base
H2O acid
HC6H6O6− conjugate acid
H3O+ conjugate base
239
views
2
rank
Related Videos
Related Practice
Showing 1 of 15 videos