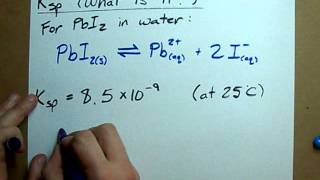18. Aqueous Equilibrium
Solubility Product Constant: Ksp
Problem 99b
Textbook Question
Textbook QuestionThe solubility of CaCO3 is pH dependent. (b) Use the Kb expression for the CO32 - ion to determine the equilibrium constant for the reaction CaCO31s2 + H2O1l2 ΔCa2 + 1aq2 + HCO3-1aq2 + OH-1aq2
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1542
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos