8. Thermochemistry
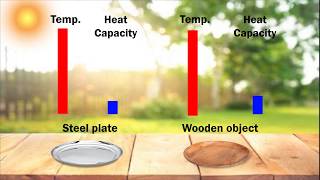
Heat Capacity
Multiple Choice
Multiple Choice50.00 g of heated metal ore is placed into an insulated beaker containing 822.5 g of water. Once the metal heats up the final temperature of the water is 32.08 ºC. If the metal gains 14.55 kJ of energy, what is the initial temperature of the water?
A
23.86 °C
B
32.86 °C
C
63.08 °C
D
36.31 °C
987
views
9
rank
3
comments
Related Videos
Related Practice
Showing 1 of 14 videos