18. Aqueous Equilibrium
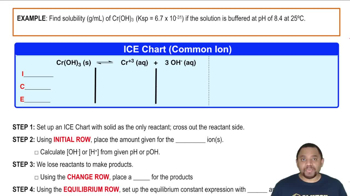
Ksp: Common Ion Effect
Problem 103
Textbook Question
Textbook QuestionThe solubility-product constant for barium permanganate, Ba1MnO422, is 2.5 * 10-10. Assume that solid Ba1MnO422 is in equilibrium with a solution of KMnO4. What concentration of KMnO4 is required to establish a concentration of 2.0 * 10-8 M for the Ba2 + ion in solution?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
940
views
Was this helpful?