15. Chemical Kinetics
Half-Life
Problem 129
Textbook Question
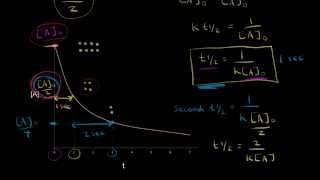
Textbook QuestionThe half-life for the first-order decomposition of N2O4 is 1.3 * 10-5 s. N2O41g2S 2 NO21g2 If N2O4 is introduced into an evacuated flask at a pressure of 17.0 mm Hg, how many seconds are required for the pressure of NO2 to reach 1.3 mm Hg?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
1170
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos