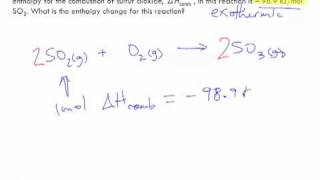8. Thermochemistry
Thermochemical Equations
Multiple Choice
Multiple ChoiceConsider the following reaction:
2 C6H6 (l) + 15 O2 (g) → 12 CO2 (g) + 6 H2O (g) ∆Hº = – 6278 kJ
What volume of benzene (C6H6, d = 0.880 g/mL, molar mass = 78.11 g/mol) is necessary to evolve 5.19 x 109 kJ of heat?
A
1.47 x 108 mL
B
5.19 x 109 mL
C
7.34 x 107 mL
D
9.51 x 109 mL
831
views
Related Videos
Related Practice
Showing 1 of 10 videos