15. Chemical Kinetics
Energy Diagrams
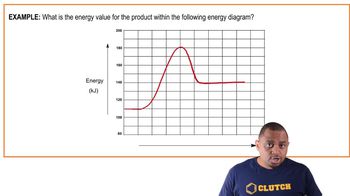
Problem 11a
Textbook Question
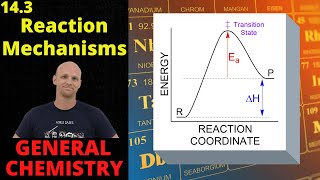
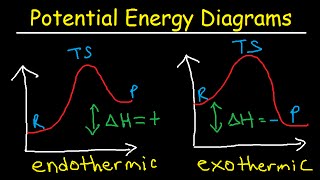
Textbook QuestionA key reaction in the upper atmosphere is O31g2 + O1g2 ¡ 2 O21g2 For this process, the energy of activation for the forward reaction, Ea1fwd2, is 19 kJ/mol, and the enthalpy change for the reaction, ΔHrxn, is -392 kJ>mol. What is the energy of activation for the reverse reaction, Ea1reverse2? (LO 14.10) (a) 411 kJ/mol (b) 392 kJ/mol (c) 373 kJ/mol (d) 196 kJ/mol
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
513
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos