10. Periodic Properties of the Elements
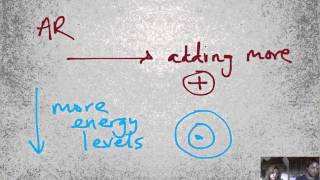
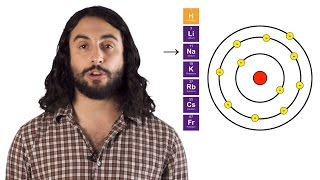
Periodic Trend: Atomic Radius
Multiple Choice
Multiple ChoiceIn moving from top to bottom in the same column on the periodic table, what trend is expected for atomic size?
A
Increase because the nucleus is getting stronger.
B
Increase because electrons are being placed in higher n-values.
C
Decrease because the nucleus is getting stronger.
D
Increase because electrons are being added to degenerate orbitals.
E
Decrease because electrons are being placed in higher n-values.
762
views
5
rank
Related Videos
Related Practice
Showing 1 of 12 videos