16. Chemical Equilibrium
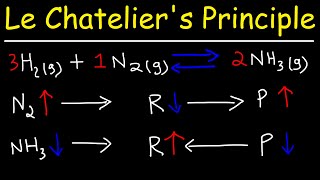
Le Chatelier's Principle
Problem 128
Textbook Question
Textbook QuestionThe following reaction is important in gold mining: 4 Au1s2 + 8 CN-1aq2 + O21g2 + 2 H2O1l2 ∆ 4 Au1CN22-1aq2 + 4 OH-1aq2For a reaction mixture at equilibrium, in which direction would the reaction go to reestablish equilibrium after each of the following changes? (a) Adding gold
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
289
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos